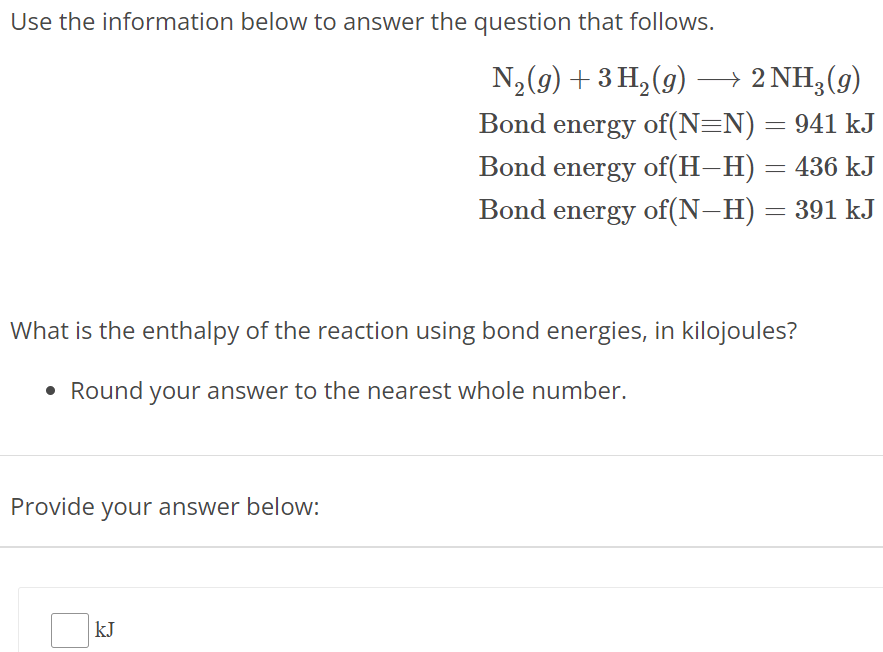
Use the information below to answer the question that follows. N2(g) + 3H2(g) ⟶ 2NH3(g) Bond energy of (N≡N) = 941 kJ Bond energy of (H−H) = 436 kJ Bond energy of (N−H) = 391 kJ What is the enthalpy of the reaction using bond energies, in kilojoules? Round your answer to the nearest whole number. Provide your answer below: kJ


You'll get a detailed, step-by-step and expert verified solution.
 Work With Experts to Reach at Correct Answers
Work With Experts to Reach at Correct Answers